Massive hype (and real hope) exists relevant Lecanemab, the newly approved (July 2023) medication for Alzheimer’s disease. Earlier amyloid-beta monoclonal antibodies largely failed but Lecanemab demonstrated a statistically significant (albeit mild) but perhaps not clinically significant reduction in cognitive decline compared with placebo (difference +0.45 points on a 18 point scale over18 months). While this is a quite mild difference in decline, the hope is that the slowing of decline will be sustained over many years making a major difference.
https://pubmed.ncbi.nlm.nih.gov/36449413/
One problem was that Lecanemab tx is associated with significant increased rate of brain shrinkage (compared to the placebo rate of shrinkage) at the recommended 10mg/kg dose.
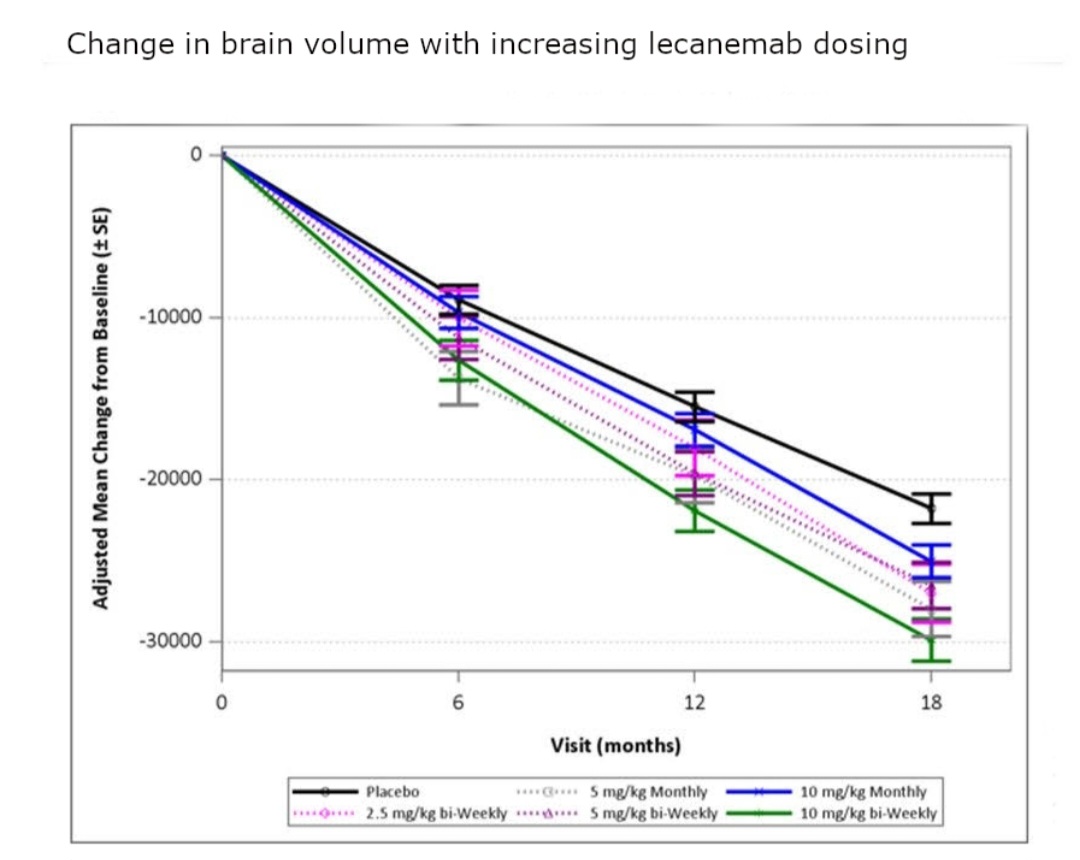
The consequence of this increased rate of brain shrinkage for the future of these patients is unknown. Trading a 0.45 point cognitive gain over placebo for an equivalent of 7 month increase in brain shrinkage compared with placebo over 18 months raises questions.
Brain shrinkage is considered to be important pathology and it has been suggested that this level of increased loss cannot be explained by Amyloid clearance. This provocative finding raises the question as to whether, notwithstanding the short term reduction in cognitive decline, Lecanemab might cause accelerated cognitive decline years later.
This month a metanalysis was published which examined lithium as an alternative as this medication may also have disease modifying effect.
One problem with the Landmark Lecanemab trial is it used change from baseline and the placebo group was slightly more sick so an ajustment was applied.
Hopefully there will be comments about these important trials.