An interesting trial found that a treatment delayed disease onset, but did not reduce overall incidence at end of follow up:
What estimand would you target in such cases?
I thought about “difference in time unwell” as in https://jamanetwork.com/journals/jamanetworkopen/fullarticle/2825081 but I wonder if there are other interesting estimands for this interesting clinical case scenario
2 Likes
I maybe misunderstanding estimands, but why would you change your estimand given the result? For Clostridium (I refuse to change the name) difficile colitis, physicians use a vancomycin taper in the hope of preventing recurrence. There is no evidence that the taper did so. I would both as a physician and potential patient consider this a failure. If anything, the taper has caused harm because it delays the offering of another hopefully more effective treatment.
2 Likes
I didn’t want to bring many clinical details because didn’t want to focus the estimand question to this disease specifically, but sure:
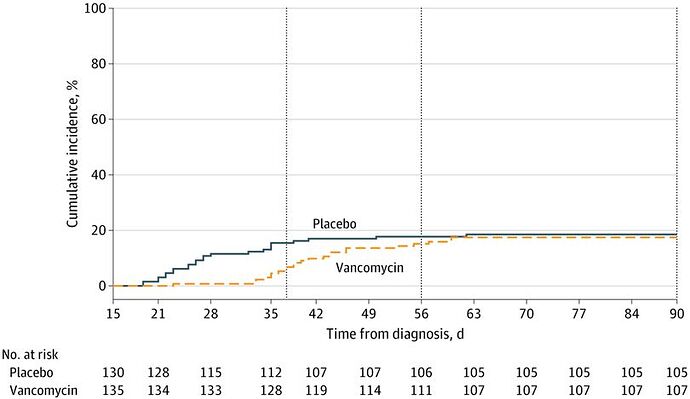
CDI has been historically treated with vancomycin for 10-14 days. A recent RCT showed that a newer drug, fidaxomicin, had less CDI recurrence (new-onset CDI after the original treatment time). However, the key point here is that fidaxomicin RCT showed better results ending the follow-up at ~38 days.
Then this newer RCT, that did not test fidamoxin but instead a taper dosage of vancomycin, ALSO showed better results of taper vanco at 38 days, but no difference later on!
Does fidaxomicin reduce CDI recurrence or just delays it, like taper vanco? No one knows.
Nevertheless, I want to explore this type of clinical scenario where a treatment doesn’t eliminate a person’s risk to a disease, but instead seems to delays its onset.
Is it a fail?
The premise that statistical method per se may substitute for particular judgments adapted to specific circumstances (like those invoked by Elias) reminds me of what Hayek identified as the ‘fatal conceit’ of central planning.
Perhaps this whole Estimands Framework thing is a fail!
I think the estimates framework is dramatically overhyped but still offers a bit. Particular judgments are key; the same as saying that a utility function needs to be considered before revealing the results. For a weight loss study the clear estimand would be the final weight, ignoring the journey. For a pain study, any day pain-free is good so using a Markov longitudinal model to estimate pain-free days or days with pain level < 2 would be appropriate. I’m not as clear on the clinical setting for the vanc study.
2 Likes